


Want to learn more about algaecide? Read on to find out when to add algaecide to your pool maintenance routine and other helpful tips.
Learn about the chemicals you use to keep your pool and hot tub water crystal clear!

In this detailed guide, we’ll help you find the best pool shock, so you can keep your chlorine or saltwater pool clean, clear, and healthy for swimming.

In this buying guide for the best pool algaecide, we’ll help you find the best product based on swimming pool type, budget, and specific needs.

Want to learn more about algaecide? Read on to find out when to add algaecide to your pool maintenance routine and other helpful tips.


There are many benefits to having a saltwater pool. However, you can’t reap the benefits without the proper equipment. Read on to learn more.


Muriatic acid is one of the most versatile pool chemicals. Read on as we answer the most common muriatic acid pool questions.


If you’re a pool or hot tub owner and are interested in the growing trend of salt water pool conversion keep reading to learn more about the process!


Trying to figure out which product to buy for your cloudy pool? Read on to find out whether a clarifier or flocculant is best for you.


Green hair from chlorine occurs because chlorine oxidizes hard metals in the pool water. Learn more about how to prevent green hair.



In this quick guide, we’ll answer the question “can you over shock a pool” and unveil the factors to consider when shocking a pool.


Saltwater generators are quickly becoming a popular sanitizing system for swimming pools and hot tubs. This article describes how they work.


The cyanuric acid level of your pool impacts the effectiveness of you chlorine. Suggested Free Chlorine takes this relationship into account.


What you sanitize your pool with is one of the most crucial decisions you will make as a pool owner. Contrary to popular belief, there are many different ways to clean your pool. Chlorine is a great and reliable option, but it isn’t the only one.


Free Chlorine and Total Chlorine mean different things for your swimming pool. Knowing the difference is key to a well balanced pool.


The Calcium Saturation Index (CSI) is a helpful tool for testing your swimming pool’s health. Read on to learn how to use it!
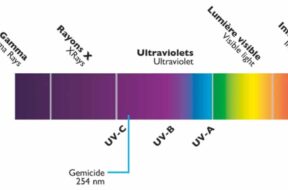

One of the latest and greatest innovations in pool technology is the use of UV rays for pool sanitation and treatment. This UV system is a supplemental pool tool that can greatly increase the cleanliness of your pool as long as it is used in an efficient way.


Borates provide many benefits to your swimming pool. Read on to learn more about why you should consider using borates!


The first chlorine pool was invented and used in the year 1911 by Brown University (See source here). Since then, chlorine pools have become the most popular type of pool in the world. But is it time to rethink that norm?


Sulfuric acid can be used to reduce the pH of your pool. But is it the right choice for you? Read on to learn more!


Shocking your pool should be part of your regular maintenance. It will help you avoid algae and bacteria build-up, and save you from cloudy water.


This article describes the chemistry involved in keeping your pool or hot tub safe and your water crystal clear.


Total alkalinity is similar and correlated to cyanuric acid. It’s not as volatile as free chlorine or pH and doesn’t require frequent testing.


You should test your pool pH level as often as you test free chlorine – about twice per week. To raise pH use borax or soda ash (aka washing soda). To lower pH use muriatic acid.
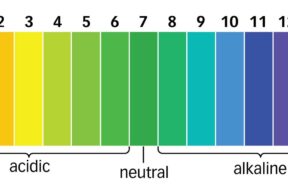

The ideal pH level in both swimming pools and hot tubs is 7.4 to 7.6. This is the level where water is most comfortable for swimmers while also maximizing chlorine’s sanitizing ability.


New to owning a swimming pool? This article explains water testing and what you need to know to keep your pool water clear and balanced.


In general, chlorine levels should be tested at least twice per week. However, the answer to this question depends heavily on how often your swimming pool is used, who uses your pool, and weather events that may introduce a large amount of fresh water into your pool.


Chlorine is the most important chemical when it comes to maintaining sparkling clean water that is safe to swim in. Chlorine sanitizes your pool water by attacking and killing harmful microorganisms, called contaminates, that could lead adverse health consequences. Therefore, it is critical to know how to test the chlorine in your pool or hot tub, as well as what to do when your chlorine levels are outside recommended ranges.
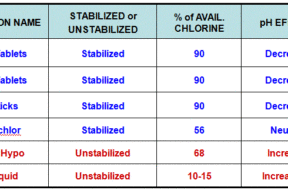

Sanitizers come in liquid, powder, and tablet form, and many pool owners have questions about the differences between them. In this article, we will get your questions answered.



The Association of Pool and Spa Professionals recommends free chlorine levels for both swimming pools and hot tubs be kept between 2.0 and 4.0 ppm. However, the Center for Disease Control recommends free chlorine stay above 1 ppm in pools and 3 ppm in hot tubs.


The suggested cyanuric acid level depends on the amount of direct sunlight your pool receives and whether you use a salt chlorine generator.


Test strips are the easiest way to test cyanuric acid. Cyanuric acid is raised by adding chlorine stabilizer containing cyanuric acid. The only way to lower cyanuric acid is by replacing water.


There are three types of pool stabilizer: stabilized chlorine, liquid stabilizer, and granular stabilizer. Which type of stabilizer you should use is largely a matter of personal preference. Learn more about pros and cons of each.


There are is a lot of misunderstanding about how chlorine works to kill harmful contaminants in pool water, as well is misconceptions about what type of contaminants chlorine will kill efficiently. In this article, we cover 5 important things free chlorine will kill in your swimming pool or hot tub water.
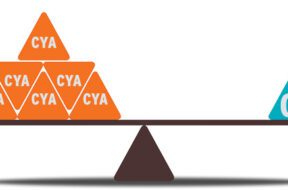

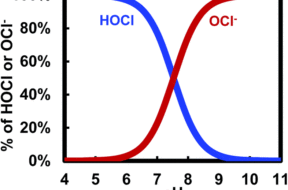
The degree to which chlorine is effective in sanitizing your pool by breaking down contaminants is highly correlated to the degree to which pH is kept in balance.
In this article, we dive into the relationship between chlorine and pH, and how pH of water changes the effectiveness of chlorine.

Learn the relationship between chlorine and cyanuric acid, and how cyanuric acid stabilizes the free chlorine in your pool.


Cyanuric acid stabilizes free chlorine by preventing it from evaporating from UV sunlight, which increases the lifespan of free chlorine but decreases its ability to sanitize swimming pool water.



Maintaining both pH and total alkalinity in your swimming pool is important for keeping your pool properly sanitized and non-corrosive. Total alkalinity is to pH what cyanuric acid is to free chlorine. Total alkalinity stabilizes pH levels. The ideal pool pH level is 7.4 to 7.6. The ideal total alkalinity level is 80 to 120 ppm.


Swimming Pool alkalinity (often called total alkalinity) is a measurement of the concentration of alkaline materials in your pool. Alkaline materials stabilize pH but buffer chlorine sanitation.


Pool algae is an unfortunate yet common problem many pool owners face. This article describes the types of algae and how you can prevent it.


Saltwater and chlorine are the two most common types of pool sanitation. Learn the benefits and challenges with both!


Want to know what the different types of chlorine mean? Read on to learn about Free chlorine, combined Chlorine, and Total chlorine.



Want to learn more about algaecide? Read on to find out when to add algaecide to your pool maintenance routine and other helpful tips.



In this quick guide, we’ll answer the question “can you over shock a pool” and unveil the factors to consider when shocking a pool.



Maintaining both pH and total alkalinity in your swimming pool is important for keeping your pool properly sanitized and non-corrosive. Total alkalinity is to pH what cyanuric acid is to free chlorine. Total alkalinity stabilizes pH levels. The ideal pool pH level is 7.4 to 7.6. The ideal total alkalinity level is 80 to 120 ppm.



The Association of Pool and Spa Professionals recommends free chlorine levels for both swimming pools and hot tubs be kept between 2.0 and 4.0 ppm. However, the Center for Disease Control recommends free chlorine stay above 1 ppm in pools and 3 ppm in hot tubs.